With more than 18,000 heath care professionals reportedly in attendance this past November in Dallas, Texas, the American Heart Association (AHA) Scientific Sessions remains the largest meeting on the science and medicine of heart disease in the world. The impact of the University of Ottawa Heart Institute continues to grow at this leading event.
Responsible once again for more than 40 program items, the Heart Institute reported on a wide range of clinical and research topics. In particular, the Heart Institute’s 2013 contributions included a large number of expert clinical talks ranging across topics from minimally invasive surgery to cardiac imaging.
Surgeon Vincent Chan, MD, spoke on appropriate long-term follow-up for mitral valve repair patients. Electrophysiologist David Birnie, MD, provided the Canadian perspective on warfarin and other new anti-coagulant medications. Chief of Cardiac Surgery Marc Ruel, MD, spoke on achieving long-term functionality of bypass grafts while reducing the risk to patients through minimally invasive techniques.
“AHA brings science and clinical practice in surgery, cardiology and nursing all together. And in terms of the science, it is the best,” said Dr. Ruel, who also serves as the AHA Surgery Scientific Program Chair. “An oral presentation or an invited talk at AHA is the height of visibility.”
Dr. Ruel indicated that the Heart Institute’s 10 expert talks and numerous research presentations are a mark of the organization’s growing international leadership. “It would not be an overstatement to say that the Heart Institute is the most prominent Canadian institution at AHA,” he said.
Along with former Heart Institute surgical resident Alex Kulik, MD, Dr. Ruel is co-chairing the development of the AHA consensus statement on the optimal treatment of patients following coronary artery bypass surgery, scheduled for release in 2014. “Equally important to the bypass surgery itself is to make sure that the disease doesn’t come back,” he explained. “The Heart Institute has done much important work in secondary prevention through our Division of Prevention and Rehabilitation.”
Best Biomaterials for Aortic Valve Repair
A variety of biomaterials are used in aortic valve repair to replace the cusps (leaflets) that let the valve close tightly after each heartbeat. But these materials, many commonly used, have never been tested head-to-head in a repaired aortic valve to see which are most likely to provide long-term durability.
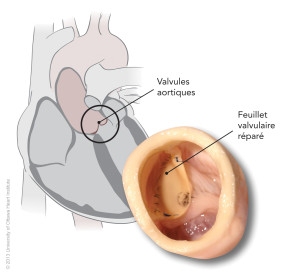
Surgical resident Hadi Toeg, MD, and a team of Heart Institute researchers have remedied this information gap with a study that brought Dr. Toeg recognition as a finalist for the AHA Vivien Thomas Young Investigator Award. They tested five biomaterials for aortic valve repair:
- Autologous porcine pericardium (APP), derived from pig tissue
- Glutaraldehyde-fixed bovine pericardial patch (BPP, Synovis™), derived from cow tissue
- Extracellular matrix scaffold (CorMatrix™), derived from pig tissue
- Collagen-impregnated Dacron (HEMASHIELD™), synthetic fiber-based material
- SJM™ Pericardial Patch with EnCap™ Technology (St. Jude Medical), derived from cow tissue
All but the SJM material, which is still experimental, are currently used for valve repair. The researchers used each material to repair an aortic valve by replacing a cusp. They then tested the repaired valves using a machine that simulates the pressure exerted by the left side of the heart. Each material was used in a repair and tested in the machine at least seven times.
Compared with intact valves, repaired valves that incorporated the HEMA-SHIELD, CorMatrix, and BPP materials registered an increase in physical stress that may make them more likely to fail over time. The researchers concluded that the APP and SJM materials most closely resemble normal aortic valves in their ability to withstand stress and strain and may be the best suited to aortic valve repair.
Best of Basic Science
In the conference’s Best of Basic Science session, Nicholas Blackburn, a doctoral student in the Cardiovascular Tissue Engineering Laboratory led by Erik Suuronen, PhD, presented work testing a way to “detoxify” the heart after a heart attack.
Tissue damaged by a heart attack can produce a toxic molecule called methyl-glyoxal, which may inhibit the heart’s ability to form new blood vessels. To see whether controlling methylglyoxal levels after a heart attack improves tissue repair, the researchers used a mouse model genetically engineered to produce an excess of glyoxalase-1 (GLO1), an enzyme that removes methylglyoxal from the heart.
They found that mice that produced extra GLO1 had a lower accumulation of methylglyoxal in the heart greater density of blood vessels in the heart, and less cardiac cell death after a heart attack compared with mice that did not have extra GLO1.
Reducing the amount of methylglyoxal in the heart after a heart attack may be a novel way to promote the organ's natural healing process.

