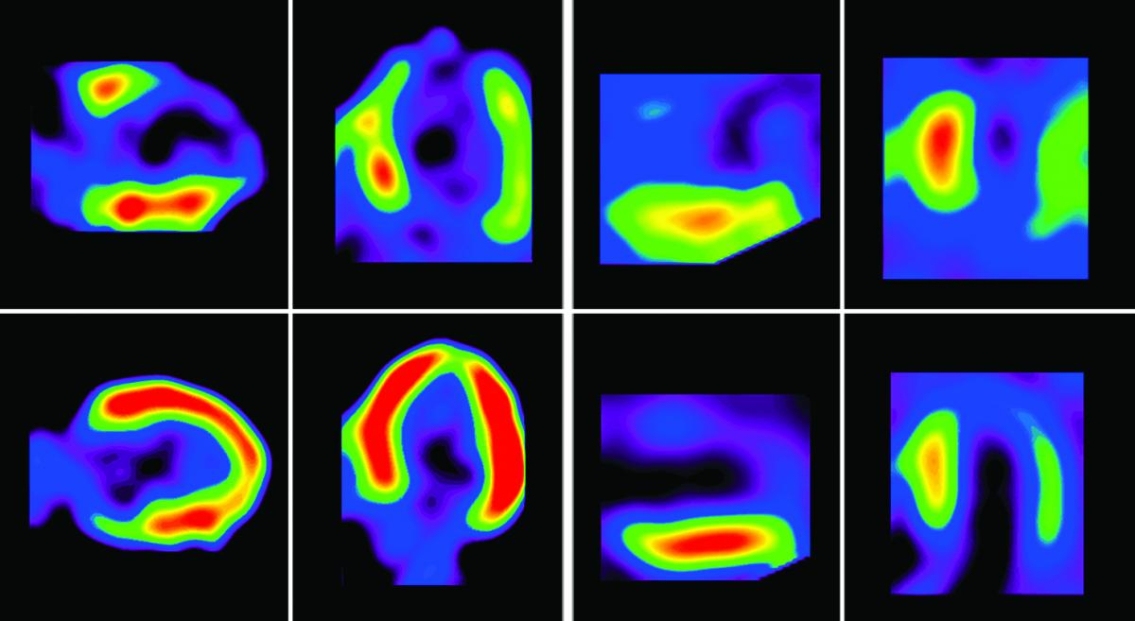
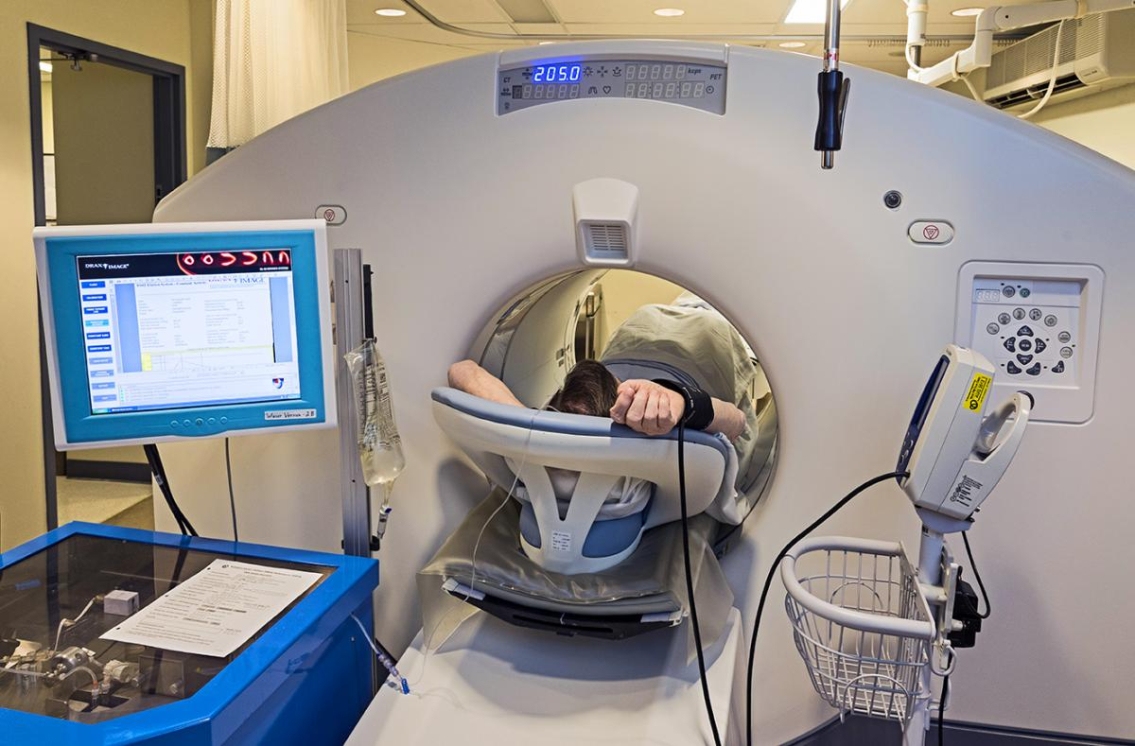
PET nuclear imaging commonly uses a radioisotope tracer with an extremely short half-life, leaving very little time to draw the tracer up into a needle and transfer it to the patient. A state-of-the-art portable technology developed by the Ottawa Heart Institute produces a personalized dose of the tracer which is infused directly into a patient lying in a PET scanner.
Recently approved by the US Food and Drug Administration (FDA), the Ruby-Fill® Generator is licensed to industry partner Jubilant DraxImage, Inc.
“The FDA approval of the Ruby-Fill® Generator is a recognition of this technology as an emerging clinical reality,” said Rob deKemp, PhD, Head Imaging Physicist at the Ottawa Heart Institute and lead developer of the portable system. “The approval will increase the use of this much-needed technology in North America and potentially worldwide through our Canadian industry partner, DraxImage.”
Cardiac PET is a non-invasive nuclear imaging procedure that measures cellular activity in the heart that would otherwise be invisible. By introducing small (trace) amounts of short-lived radioactive dye into a patient’s bloodstream, cardiologists can see in real-time through myocardial perfusion imaging what parts of the heart are using certain substances and how well that patient’s heart is functioning.
“There is a big push in the medical imaging community to image wisely and gently,” said Dr. deKemp. “With its advanced weight-based dose accuracy, among other capabilities, the Ruby-Fill® technology really is a way to help tailor the test to each patient.”
“The Ottawa Heart Institute has been a leader with this imaging approach for several years and has helped to developed clinical standards around the world,” said Rob Beanlands, MD, Division Head of Cardiology. “The Ruby-Fill® Generator is a Canadian contribution toward providing innovative, accessible technology for PET imaging.”
Coronary artery disease affects an estimated 1.3 million Canadians. With commercial launch plans expected in the next few months, the made-in-Canada generator is set to enhance the way patients with known or suspected coronary artery disease are diagnosed and managed in North America and around the world.
New PET Imaging Tracer Looks Promising for Use in Exercise Stress Testing
The use of PET imaging has not been practical in exercise stress testing for coronary artery disease because of the short half-life of the radioactive tracers used. Findings of an international Phase 3 clinical trial recently presented by Dr. Beanlands at the American Society of Nuclear Cardiology Annual Meeting, suggest that a breakthrough may be just around the corner.
With a 110-minute half-life, preliminary results show that a tracer called flurpiridaz F 18 not only makes such testing possible, but provides better diagnostic performance and sensitivity than standard SPECT imaging while reducing radiation exposure for patients. If the second Phase 3 trial—needed to apply for FDA approval—continues to demonstrate the new tracer’s accuracy, flurpiridaz F 18 is a potential game-changer in the non-invasive evaluation of heart disease.

