A class of anti-cholesterol drugs called PCSK9 inhibitors has been in the press lately, and for good reason: they can reduce blood cholesterol levels substantially beyond what a statin alone can achieve. Two have made it to market so far for treating coronary artery disease, with more likely to come.
However, the currently available inhibitors must be given by injection instead of pill form, because the molecules are large and unable to pass through the gut. They are also very expensive. Their use so far has been restricted to people with a genetic condition that leads to extremely high blood levels of LDL—the so-called “bad cholesterol.”

Thomas Lagace, PhD, director of the Lipoprotein Receptor Biology Laboratory at the Ottawa Heart Institute, has been at the forefront of PCSK9 research for his entire career. At the America Heart Association 2016 Scientific Sessions, he gave a presentation called “Emerging Mechanisms of PCSK9 Lowering of LDL Cholesterol: Beyond the LDL Receptor.” By better understanding how PCSK9 works, his lab hopes to develop better and cheaper ways to block it.
PCSK9 makes a great target for cholesterol lowering, explained Dr. Lagace, since it’s likely that the modern human body has no need for it. In early work as a postdoctoral fellow, Dr. Lagace and his colleagues found that PCSK9 binds to and marks for destruction the cellular receptors for LDL cholesterol in the liver. When levels of this LDL receptor drop, blood cholesterol levels rise.
Later work from his group at the Heart Institute showed that PCSK9 can also bind to LDL circulating in the bloodstream, and when bound this way, can no longer destroy the LDL receptor.
These two mechanisms are likely part of an ancient feedback loop that kept certain lipids in the circulation as long as possible, delivering much-needed fuel to the heart and muscles, explained Dr. Lagace.
“These mechanisms evolved at a time when food was scarce, and you might go a long time fasting. But a modern diet is quite incompatible with this,” he said. “We’re constantly producing PCSK9, and it’s just getting in the way and preventing LDL from being cleared by the liver.”
His lab has been trying to figure out exactly how circulating LDL binds to PCSK9 and shuts it down. “If we can tap into that down-regulation mechanism, we may be able to mimic it using a small molecule drug or some other type of therapeutic,” he said.
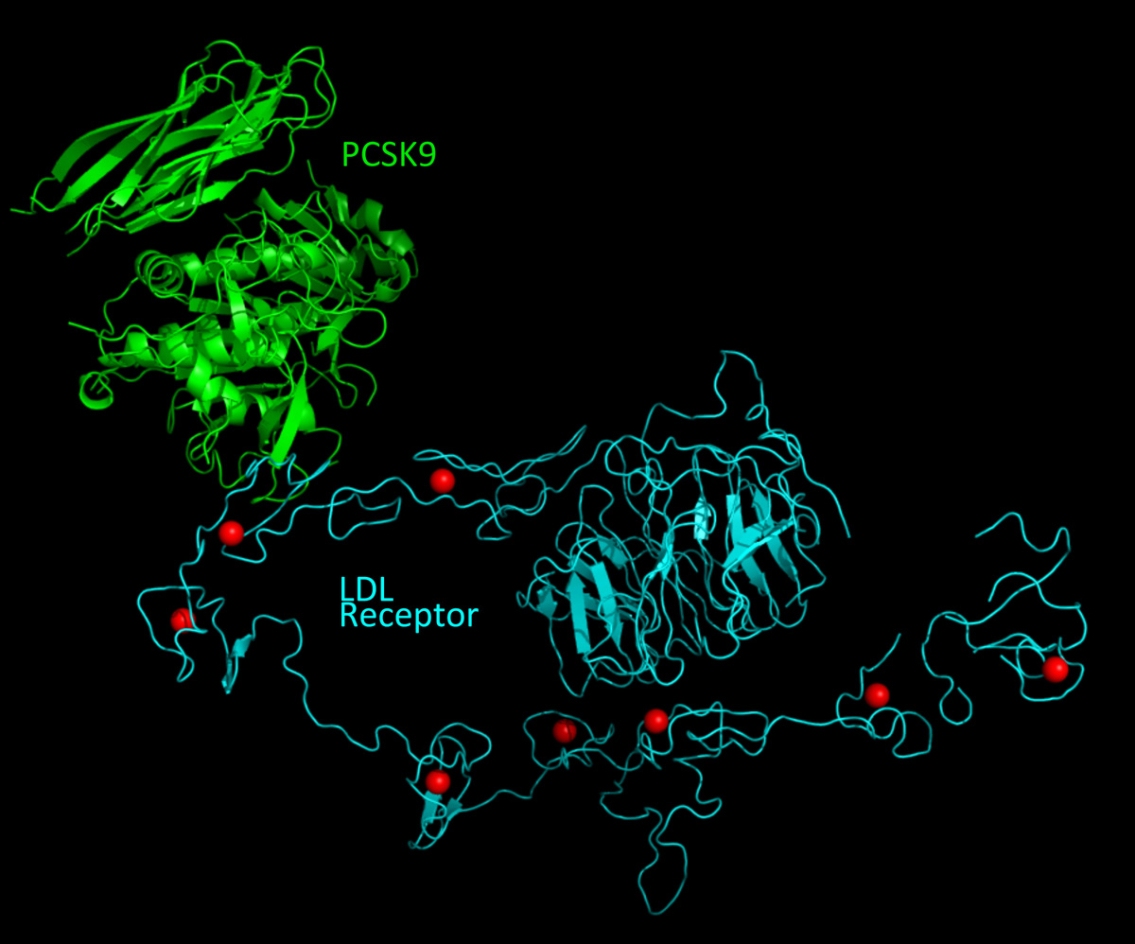
Several years ago, Dr. Lagace and his colleagues made an unexpected discovery while using X-ray crystallography to determine the molecular structure of the part of PCSK9 that binds to the LDL receptor. They cut off a small piece of the protein far away from that site, which was getting in the way of their image. To their surprise, making this cut altered the protein so that it bound better to the LDL receptor. Adding to the intrigue, they recently found the same altered PCSK9 protein could no longer bind to LDL.
Dr. Lagace and his team are now working to create a mouse model with a similar loss of function, so they can test in detail how exactly LDL binding inhibits PCSK9 activity.
He explained that this target area on the protein is very small. “If we learn more about this mechanism, then we will have found another way to inhibit PCSK9. That might even be something that could be given in a pill. That would make PCSK9 inhibition more accessible to more people,” he added.
Early results of a clinical trial of one of the approved PCSK9 inhibitors presented at AHA 2016 generated great excitement, showing that patients’ cholesterol levels dropped much lower than seen with a control group receiving statin drugs alone. Correspondingly, atherosclerotic lesions actually shrunk in size.
“The search for new inhibitors of PCSK9 is very important, because these results are just so dramatic. There was talk at the meeting that we may see the end of coronary artery disease if we can make PCSK9 inhibitors accessible,” concluded Dr. Lagace. “There seems to be no limit to how low you can take LDL cholesterol and still see further benefit.”

