Somewhere between traditional cardiologists and cardiac surgeons sit interventional cardiologists. Like surgeons, interventionists get blood on their gowns in the process of correcting structural problems of the heart, but their approach is more subtle and less invasive. Spending their days in the catheterization lab instead of the operating room, their tool is the catheter rather than the scalpel.
“I think the seven of us are just driven,” offered Dr. Marino Labinaz as he described the interventional cardiologists at the University of Ottawa Heart Institute. Dr. Labinaz is the Director of the Cardiac Catheterization Laboratory and leads a team that treated close to 2,500 patients 2011 to 2012. In fact, the team performs more procedures each year than other academic hospitals with almost double the Heart Institute’s interventional staff.
The majority of that work involves standard coronary catheterization to identify blocked or narrowed arteries and inserting stents in those arteries to open them and return blood flow to the heart muscle. But it is not just productivity that drives the team. “You could say we innovate as well as treat,” said Dr. Labinaz. Each of the seven is also champions of new, specialized procedures and technologies that have the potential to change cardiac care and offer hope to patients who may have no alternatives available. “Interventional cardiology took off in the 1980s with the introduction of angioplasty, and there’s been a great race to find new ways of treating heart disease without surgery,” added Dr. Christopher Glover. “We’re always working on ways to make things a little less invasive, with less trauma to patients so they can recover more quickly.”
Correcting Defective Valves Without a Scalpel
Dr. Labinaz has led the effort to bring two catheter-based heart valve procedures to the Heart Institute. Each deals with a different valve but both offer a treatment option to patients for whom surgery presents too great a risk. These procedures involve close collaboration between cardiology and Surgery, from patient selection through to recovery.
Aortic stenosis—narrowing of the aortic valve—affects nearly 200,000 Canadians and, left untreated, can lead to debilitating circulation problems and death. Many sufferers are elderly and not good candidates for open heart surgery. Transcatheter aortic valve implantation (TAVI) offers a non-invasive way to implant a new aortic valve. With the replacement valve folded onto a catheter, it is inserted in to the femoral artery in the groin, threaded into position in the heart and expanded into place. (See gallery)
A Stent That Won’t Overstay Its Welcome
A coronary stent is a wire mesh tube used to prop open a blocked or narrowed artery. Opening closed arteries by inserting stents through catheters is known as percutaneous coronary intervention (PCI). It is the bread and butter of interventional cardiology.
So-called “bare metal” stents debuted in the mid-1980s and are plain stainless metal. Starting in the early 2000s, drug-eluting stents began to appear. They are coated with material that slowly releases drugs to prevent cells and clots from forming at the repair site that could reblock the artery. This is an improvement, but the risk of reclosure, or restenosis, remains over the long term.
A fully absorbable stent has many theoretical advantages, explained Dr. Labinaz, who first worked on the stent conceptually as a fellow almost 20 years ago. “The healing response to stents really happens in the first six months, and the bioabsorbable scaffolds start dissolving at six to nine months.” He is now participating in the ABSORB-EXTEND trial, which is testing whether a completely bioabsorbable stent can provide better long-term healing for blood vessels.
“After many years, atherosclerosis can begin to build up in stented arteries,” he continued. “So we’re hoping that, by having the artery return to its normal state, we can reduce that risk. And we now know that when the stent absorbs completely, the artery returns to its normal biological functioning—it relaxes, it constricts—and that may be a good thing down the road.” The first patients in the trial at the Heart Institute received the new devices in June of this year.
“I predict that in five to 10 years, the majority of tissue aortic valves will be placed by TAVI, and it will be very uncommon to do surgical placement,” said Dr. Labinaz. He and surgeon Dr. Marc Ruel began the TAVI program in 2007. With Dr. Glover and surgeon Dr. Buu-Khanh Lam, they are now participating in the international SURTAVI clinical trial to compare TAVI with open heart surgery as a solution for patients at intermediate risk of complications with standard valve surgery. TAVI has already become standard for high-risk or inoperable patients.
Dr. Labinaz is one of the first Canadian cardiologists to employ a device called the MitraClip to repair the mitral valve, in collaboration with Chief of Surgery Dr. Thierry Mesana and anesthesiologist Dr. Mark Hynes. Advanced heart failure can enlarge tissue around the valve and prevent it from closing properly. The valve itself is often healthy, but the leaflets that open and close to control blood flow no longer form a tight seal. The backflow of blood with each beat of the heart leads to decreased circulation through the body and severe shortness of breath.
Like TAVI, the MitraClip procedure is a true multidisciplinary effort, requiring collaboration from surgeons and heart failure specialists who help pick the most appropriate patients, and anesthesiologists who perform the transesophageal echocardiography that provides the visuals for the tricky manoeuver of clipping the leaflets while the heart continues to beat.
The team has so far performed MitraClip repairs in four patients, each with excellent results. “These patients all had severe regurgitation. On a scale of 0 to 4, they all had 4, and we reduced them to 1,” said Dr. Labinaz, a level where they are functional again.
Another catheter-based approach—mitral valve implantation, where a new valve is sewn on top of the old leaky one, like a tooth being capped—has recently been performed in Europe for the first time. “Whether the MitraClip is the definitive therapy for this type of defect, I’m not sure. But it will still have a niche because it does help certain patients with a certain type of problem. It’s better to have more options so that we can treat more people,” he concluded.
Personalized Drug Therapy for Stent Patients
Patients who have had stents inserted to open blocked arteries must take anti-clotting medication for at least a year. The most widely used drug is clopidogrel, but some individuals do not have a proper response to it. To identify these patients, Dr. Derek So has brought personalized genomics to interventional cardiology and, in the process, has led the development of RAPID GENE, the first-ever point-of-care genetic test in medical history.
In a paper published in May of 2012 in The Lancet, he and his colleagues showed that a bedside test for a common genetic variation can identify angioplasty patients who will not respond to clopidogrel properly. Having this information in hand allowed doctors to give these patients an alternative drug, reducing the risk of clotting after therapy.
Dr. So’s team showed that by using a genetic test to direct anti-clotting therapies, “you could actually prevent more bleeding outcomes, and at the same time also decrease costs by directing the right medication to the right people,” he said Dr. So. Full details about the RAPID GENE study can be found in Volume 7, Issue 1, and Volume 7, Issue 2, of The Beat.
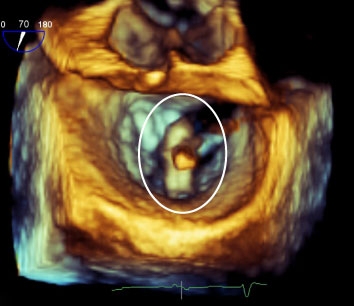
Putting a Plug in Clot-Prone Hearts
Atrial fibrillation is a type of irregular heartbeat common in elderly patients and the most common arrhythmia overall. The condition can disrupt blood flow through the heart and increase the risk of blood clots and stroke. For patients unable to take anti-clotting medication, a new catheter-based device is showing great promise in preventing clot formation, said Dr. Jean-François Marquis. “As the population ages, this is becoming more of a problem.”
With atrial fibrillation, blood can pool and clot in a small pouch in the front of the heart called the atrial appendage. Most patients with atrial fibrillation can take blood thinners, such as warfarin, but some cannot due to the risk of dangerous bleeding in the brain, gut or elsewhere.
A new solution being tested in Canada under a compassionate grant is called an atrial appendage closure device. “What we do,” explained Dr. Marquis, “is put a catheter inside that appendage and insert the device—a cylinder with hooks. It attaches there and blocks the appendage off. The device becomes covered with tissue over a period of three to five months. If there are clots in there, they’ll stay there and no new clots will be able to form.”

The procedure takes a path through the heart that requires great precision. The catheter must be pushed through the septum—the wall between the chambers of the heart. Dr. Marquis has been using this approach for almost 25 years for other interventions. “I’m one of the people here with the most experience with the trans-septal approach,” he said.
Dr. Marquis is one of only a handful of interventionists currently performing the closure in Canada, but he sees potential for great expansion if a planned clinical trial shows the procedure to be as effective as standard blood thinners for reducing stroke. The drugs are not expensive, but they must be taken for life and are not without side effects. With the lifetime risk of developing atrial fibrillation estimated to be about 20 per cent, closure of the atrial appendage could well become much more common.
Into the Cath Lab and into the Cold for High-Risk Heart Attacks
Process is often just as important as technology in improving medical outcomes, and the interventional cardiology team does not limit its innovation to devices. The Heart Institute’s groundbreaking STEMI program is a case in point. Pioneered by Dr. Michel Le May, it has reduced deaths in the region from the most dangerous kind of heart attack—ST-elevated myocardial infarctions (STEMIs)—by half since its inception in 2004 and has been adopted by institutions around the country.
High-Proof Treatment for Enlarged Hearts
Dr. Glover also specializes in ethanol ablation—a catheter-based procedure to treat a type of thickening of the heart muscle called chronic obstructive hypertrophic cardiomyopathy. Left untreated, the condition can lead to heart failure.
The traditional surgical approach, if the patient becomes symptomatic, is to open the chest, with the patient on a heart-lung machine, and cut out the excess muscle. While effective, “such invasive surgery isn’t suitable for everybody,” he explained.
Dr. Glover introduced ethanol ablation to the Heart Institute in collaboration with fellow cardiologists Drs. Kwan-Leung Chan and Luc Beauchesne after training at Baylor University in Texas. The procedure involves threading a catheter to the septum—the muscle between the chambers of the heart. The enlarged tissue is doused with alcohol, causing tiny, localized heart attacks. These tiny heart attacks scar and kill the excess muscle, relieving the physical obstruction blocking blood flow.
While the procedure is only appropriate for certain patients, “It’s a great innovation,” said Dr. Glover. “About 95 of my patients have had significant symptom relief. Most of them are very happy that they had the procedure and that they didn’t have to go for more invasive surgery.”
The STEMI protocol is all about diagnosing patients in the field, getting them directly to the cath lab and immediately stenting them to get blood flowing to the heart as soon as possible. “We had to get the emergency room physicians empowered to make decisions about care; we had to get ECGs on the ambulances; we had to get paramedics to bring patients directly to the Heart Institute from the field; we had to have one phone number to call,” recalled Dr. Le May.
“These are all logistical issues that we resolved, and we were the first to show that it’s doable and to publish on our citywide program in the New England Journal of Medicine,” he continued. Since then, the citywide program he founded has expanded to the entire eastern Ontario region. “I’ve gone around the country coast to coast, talking about our program and what we do. And people have brought it home and done what they could do with their own environment and resources,” added Dr. Le May.
“We’re always working on ways to make things a little less invasive, with less trauma to patients so they can recover more quickly.”– Dr. Christopher Glover Interventional Cardiology, UOHI

The STEMI team has now gone a step further. Building on the infrastructure it put together for “code STEMI,” the team is working on what it call “code ROSC,” a new program to care for heart attack patients who remain comatose after circulation is restored in the field. ROSC stands for “return of spontaneous circulation,” and the goal for these patients is to bring their body temperatures as low as 33 degrees Celsius as quickly as possible. (Normal body temperature is 37 degrees Celsius.)
These patients are at very high risk of brain damage or death. “There’s been research showing that if you cool the brain, cool the patient, you have a better survival rate, and you have a better neurological outcome,” explained Dr. Le May.
In the new code ROSC protocol, comatose patients are routed immediately to the cath lab at the Heart Institute, where a rapid heat exchange system cools the patient’s whole body by 4 degrees Celsius within two hours.
Code ROSC requires sophisticated protocols and collaboration among many specialties, including Emergency Department physicians, cardiologists, anesthesiologists, infectious disease specialists (to deal with pneumonia and other respiratory complications), kidney specialists, neurologists and others.
“The Heart Institute is a leader in this team approach, making sure that all the specialties are involved in managing these patients. Like the STEMI program, this code ROSC program was developed in conjunction with the hospitals around Ottawa, and the plan is to gradually expand so the pathways and protocols will appeal and apply to the entire region,” said Dr. Le May.
Offering Everything They Can
Despite the challenges and workload, the interventional team has no wish to stop innovating. The opportunity to expand and shape patient care is part of the attraction of working in a specialized cardiovascular centre. The team hopes to recruit two additional physicians over the next two years to expand the program.
“It’s exciting to be part of these new procedures, and it’s exciting to be able to offer them to patients, especially if we get such great outcomes from them,” concluded Dr. Glover. “I think we’re driven by the desire to be at the cutting edge and being able to offer everything we can to the patients in our area and across Canada.”
Opening a Back Door to Chronically Occluded Arteries
Sometimes, standard angioplasty just is not enough to restore blood flow. When an artery has been blocked for a long time, the plaque deposit can be so tough and resistant that a catheter cannot always be successfully guided through it in the standard way—in the same direction as the blood flows. Opening these chronically occluded arteries requires an advanced technique with specialized catheters and a tricky pathway through the heart.
When a coronary artery is chronically blocked, the heart can develop small, fragile blood vessels, called collaterals, which try to move blood around the obstruction. Dr. Alexander Dick has become an expert in a backdoor technique that takes advantage of these access points to reach the plaque from the other side. The catheters have a slippery coating that allows them to slide through the delicate collaterals without causing damage.
“This approach is more advantageous because the distal end of the blockage is more easily crossed, and the ability to go backwards with the wire then allows us to come all the way back through the blocked artery,” explained Dr. Dick.
He learned the procedure in the U.S. and brought it with him when he joined the University of Ottawa Heart Institute. He hopes to train his colleagues in the technique as demand for it grows. “Opening chronically occluded arteries requires more expertise in using catheters and newer devices than standard angioplasty does,” he added. So he sees opportunity for expansion of its use in a specialized centre like the Heart Institute.
The procedure has a success rate of more than 95 per cent, compared with about 75 per cent for standard angioplasty. It may be particularly helpful for people with a single blocked artery whose angina (chest pain) is not helped by medication. These patients would not normally be candidates for bypass surgery, which is reserved for people with multiple blocked vessels. “Usually, those patients would just suffer with angina,” said Dr. Dick. “So this provides a great improvement to their quality of life.”